By Cinzia Benincasa
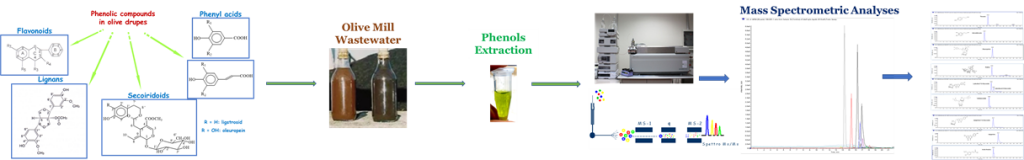
The processing of olives for oil production generates the most abundant agro-industrial by-products in the Mediterranean area (1). The production, the type, and the quantity of these by-products depend on the extraction system used for the extraction of the oil, which can be in two- or three-phase mills. The main features of the three-phase extraction system are the need to add a large amount of hot water into the decanter, up to 40 l for every 100 kg of olives processed, to dilute the olive paste, and the production of a severe quantity of vegetation water, up to 100 l for every 100 kg of olives processed (2, 3). Olive mill wastewater contains high quantities of sugars, tannins, phenolic compounds, polyalcohols, pectins, and lipids, which is highly acidic and toxic for plants (4–7). Among these, phenols, produced by the secondary metabolism of plants, represent a heterogeneous group of natural substances. Concerning the chemical diversity that characterizes them, phenols play important roles in nature: they act as a defense from herbivorous animals (they give an unpleasant taste) and from pathogens (phytoalexins); offer mechanical support (lignins) and barrier against microbial invasion; act as attractors for pollinators (anthocyanins); and as growth inhibitors of competing plants (2, 3, 6–11). The health–nutritional effects of phenols have been amply demonstrated and its intake varies enormously in relation to the type, quantity, and quality of the vegetables consumed (12–16). Since there are many benefits that phenols bring, it is worth including them in supplements (17). A rich source of bioactive substances with various biological properties that can be used as ingredients in the food industry for obtaining functional and nutraceutical foods as well as in the pharmaceutical industry is, exactly, the olive mill wastewaters, that represents a fine and cheap source of bioactive compounds that positively affect the economic status of olive companies while reducing the negative impact of olive by-products on the environment. To verify and determine the phenolic content in olive mill wastewater, spray drying methodology has been employed to obtain a powder which has been extracted for phenolic evaluation and quantitation. High-pressure liquid chromatography–tandem mass spectrometry was the technique employed to determine phenols, such as apigenin (9,55 mg/kg), caffeic acid (2,89 mg/kg), catecol (6,12 mg/kg), p-cumaric acid (5,01 mg/kg), diosmetin (3,58 mg/kg), hydroxytyrosol (1481 mg/kg), hydroxytyrosyl oleate (564 mg/Kg), luteolin (62,38 mg/kg), luteolin-7-O-glucoside (88,55 mg/kg), luteolin-4-O-glucoside (11,48 mg/kg), oleuropein (103 mg/kg), rutin (48,52 mg/kg), tyrosol (2043 mg/kg), vanillin (27,70 mg/kg) and verbascoside (700 mg/kg).
Read more at: https://doi:10.3390/molecules24193481; https://doi:10.3389/fnut.2021.782693; https://doi.org/10.3390/molecules26144337; Food Chemistry 279: 105–113.

Cinzia Benincasa obtained his degree in Chemistry in 2002 and his Ph.D. in “Methodologies for the development of molecules of pharmacological interest” at University of Calabria (Italy) in 2004. Currently, she is working as researcher for the Council for Agricultural Research and Economic (CREA) at the Research Centre for Olive, Fruit and Citrus Crops located in Rende (CS) (Italy). Her research has included: olive oil quality, food safety, traceability, trace elements, wastewaters, olive oil by-products, chemiometrics.
References
- Neifar M, Jaouani A, Ayari A, Abid O, Ben H S, Boudabous A, et al. Improving the nutritive value of olive cake by solid state cultivation of the medicinal mushroom Fomes fomentarius. Chemosphere. (2013) 91:110–4. doi: 10.1016/j.chemosphere.2012.12.015
- Obied HK, Allen MS, Bedgood DR, Prenzler PD, Robards K, Stockmann R. Bioactivity and analysis of biophenols recovered from olive mill waste. J Agric Food Chem. (2005) 53:823–37. doi: 10.1021/jf048569x
- De Marco E, Savarese M, Paduano A, Sacchi R. Characterization and fractionation of phenolic compounds extracted from olive oil mill wastewaters. Food Chem. (2007) 104:858– 67. doi: 10.1016/j.foodchem.2006.10.005
- Cassano A, Conidi C, Giorno L, Drioli E. Fractionation of olive mill wastewaters by membrane separation techniques. J Hazard Mater. (2013) 248–9:185–93. doi: 10.1016/j.jhazmat.2013.01.006
- Oreopoulou V, RussW. Utilization of By-Products and Treatment of Waste in the Food Industry. Springer (2007).
- Galanakis CM. Recovery of high added-value components from food wastes: conventional, emerging technologies and commercialized applications. Trends Food Sci Technol. (2012) 26:68–87. doi: 10.1016/j.tifs.2012. 03.003
- Allouche N, Fki I. Toward a high yield recovery of antioxidants and purified hydroxytyrosol from olive mill wastewaters. J Agric Food Chem. (2004) 52:267–73. doi: 10.1021/jf034944u
- Engin SP, Mert C. The effects of harvesting time on the physicochemical components of aronia berry. Turkish Journal of Agriculture and Forestry. (2020) 44:361–70. doi: 10.3906/tar-1903-130
- Kaskoniene V, Bimbiraite-Surviliene K, Kaskonas P, Tiso N, Cesoniene L, Daubaras R, et al. Changes in the biochemical compounds of Vaccinium myrtillus, Vaccinium vitis-idaea, and forest litter collected from various forest types. Turkish Journal of Agriculture and Forestry. (2020) 44:557–66. doi: 10.3906/tar-1912-41
- Vougogiannopoulou K, Angelopoulou MT, Pratsinis H, Grougnet R, Halabalaki M, Kletsas D, et al. Chemical and biological investigation of olive mill wastewater–OMWW secoiridoid lactones. Planta Med. (2015) 81:1205–doi: 10.1055/s-0035-1546243
- Zoidou E, Agalias A, Magiatis P, Skaltsounis AL. New iridoid derivatives from table olives cv Throuba Thassos and olive mill wastewaters. Planta Med. (2008) 74:B168. doi: 10.1055/s-0028-1084512
- Visioli F, Romani A,Mulinacci N, Zarini S, Conte D, Vincieri FF. Antioxidant and other biological activities of olive mill wastewater. J Agric Food Chem. (1999) 47:3397–401. doi: 10.1021/jf9900534
- Capasso R, Cristinzio G, Evidente A, Scognamiglio F. Isolation spectroscopy and selective phytotoxic effects of polyphenols from vegetable wastewaters. Phytochemistry. (1992) 31:4125–8. doi: 10.1016/0031-9422(92)80426-F
- Visioli F, Vinceri F, Galli C. Wastewaters from olive production are rich in natural antioxidants. Experientia. (1995) 51:32–4.
- Visioli F, Poli A, Galli C. Antioxidant and other biological activities of phenols from olives and olive oil. Med Res Rev. (2002) 22:65–75. doi: 10.1002/med.1028
- Jerman Klen T, Mozeti?c Vodopivec B. Ultrasonic extraction of phenols from olive mill wastewater: comparison with conventional methods. J Agric Food Chem. (2011) 59:12725–31. doi: 10.1021/jf202800n
- Cepo VD, Radi´c K, Jurmanovi´c S, Jug M, Grdi´c Rajkovi´c M, Pedisi´c S, et al. Valorization of olive pomace-based nutraceuticals as antioxidants in chemical, food, and biological models. Molecules. (2018) 23:2070. doi: 10.3390/molecules23082070

