By Barbara Simonato, Marilinda Lorenzini, Giacomo Zapparoli
Botrytis cinerea and Penicillium spp. (e.g. P. expanusm and P. crustosum) are the most common pathogenic agents of wine grapes (Rousseaux et al., 2014). These fungi are responsible for the production of volatile compounds that may have strong impact on the quality of wine (La Guerche et al., 2007).
Botrytis and Penicillium infect grapes are used for passito wine production during the post-harvest dehydration process (withering) (Lorenzini et al., 2016a). The incidence of their infection on withered grapes depends on culture management, seasonal conditions and withering technique. Different studies were performed to assess the B. cinerea, Penicillium spp. effects on volatile compound profile of Amarone wine, the most economically important Italian passito wine (Fedrizzi et al., 2011; Tosi et al. 2012; Zapparoli et al, 2018). Moreover, through proteomic analyses, study focused on the detection of potential markers of these fungi infection in grapes during the withering have been performed (Lorenzini, et al., 2016b; Lorenzini, et al., 2015).
In our studies, Corvina grapes were infected with B. cinerea and two Penicillum species (P. expansum and P. crustosum) in a fruit drying room of Valpolicella area. Grape samples were withered in natural conditions for about three months according traditional withering protocol. At the end of the withering process, bunches were crushed separately to obtain the grape musts for the red vinification. Must obtained from healthy Corvina grapes was used as control. Aroma compound analysis in wines obtained from healthy and infected grapes was carried out by Gas Chromatography – Mass Spectrometry (GC-MS). Sensory analysis of wines were carried out by a panel test composed by experts of Amarone wine.
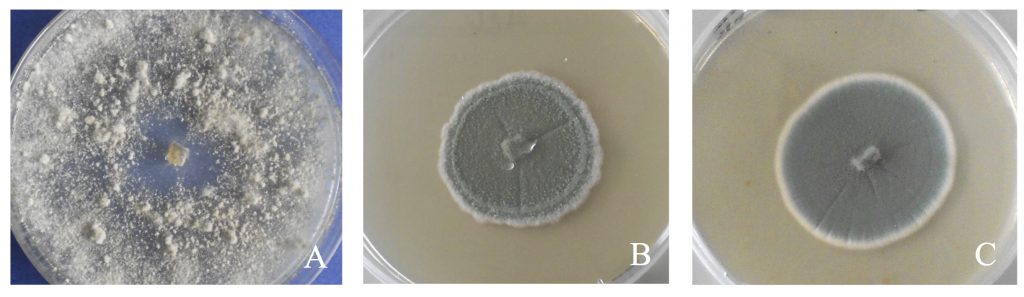
Botrytis cinerea infection of withered grapes had significant effects on Amarone wine quality. Significant changes on the content of important molecule categories such as esters, fatty acids, aldehydes and lactones were described. Similar results were reported in our previously investigations (Fedrizzi et al., 2011; Tosi et al. 2012). In particular, ethyl hexanoate, ethyl octanoate and ethyl decanoate, that play a key role in the fruity notes of wines, decreased in botrytized. Other odour-active molecules such as 1-octen-3-ol (mushroom note), phenylacetaldehyde (correlated with an oxidation-related sensory note), γ-nonalactone (coconut, sweet note) and 4-carbethoxy-γ-butyrolactone (sweet, coconut note) and N-(3-methylbutyl)-acetamide (vinegar/pungent note) increased in botrytized wine.
At sensory level, botrytized wines, tasted after one year, were perceived for more aroma complexity, almond, balsamic and resinous scent than control wine. Nevertheless, two years–aged botrytized wines were characterized by aged aroma and altered colour due to accelerated oxidation process.
Chemical and sensory analysis of wine produced from grape infected by P. expansum and P. crustosum evidenced that the fungal effects on wine quality can be important, as previously reported by Zapparoli et al. (2018). In particular, the most important odour-active molecules that varied consistently were found 3-methylthio-1-propanol (potato note), ethyl acetate (vinegar note) benzaldehydes (almond-like note), furfural (sweet, caramel scent), 4-carboxyethoxy-γ-butyrolactone, sherry lactones (oxidized note) and N-(3-methylbutyl)-acetamide. High variations in the content of these and other molecules between P. expansum and P. crustosum wines were also noted, evidencing species-related differences.
At sensory level, wine from Penicillium infected grapes were well distinguished from control wines particularly due to high intensity of pharmaceutical note giving an unpleasant aroma.


Barbara Simonato took a degree in Biological Sciences with a specialization in Food Science, at the University of Padua (Italy). She held a Ph.D. for studying the effect of technological treatment on allergenity of wheat, at the University of Padua. She is an Assistant Professor in Food Science and Technology, at the Department of Biotechnology of the University of Verona. Her research concerns the biochemistry of processed foods and their starting material.

Marilinda Lorenzini is a Ph.D. in Microbiology, at the Dipartimento di Biotecnologie of the Università degli Studi di Verona (Italy). Her research concerns the physiological, molecular and phylogenetic study of fungi (i.e. filamentous fungi and yeasts) and bacteria populations involved during the production of wines. Her research has also been concern the study of proteomic effect of filamentous fungi on grape proteome during the post-harvest withering process of grapes.
Giacomo Zapparoli is a microbiologist expert of microorganisms of grape and wine. He is an Assistant Professor in Agricultural Microbiology, at the Department of Biotechnology of the University of Verona. His research concerns the identification and taxonomy of fungi and bacteria and their effects on grape-wine quality.
Those interested in a longer length report can download the working paper at:
https://www.sciencedirect.com/science/article/pii/S0308814618307507
References:
- Fedrizzi, F., Tosi, E., Simonato, B., Finato, F., Cipriani, M., Caramia, G., & Zapparoli, G. (2011). Food Technology and Biotechnology, 49, 529−535.
- La Guerche, S., De Senneville, L., Blancard, D., & Darriet, P. (2007). Antonie Van Leeuwenhoek International Journal, 92, 331-341.
- Lorenzini, M., Millioni, R., Franchin, C., Zapparoli, G., Arrigoni, G., & Simonato, B. (2015). Food Chemistry, 179. 170–174.
- Lorenzini, M., Cappello, M.S., Logrieco, A., & Zapparoli, G. (2016a). International Journal of Food Microbiology, 238, 56-62.
- Lorenzini, M., Mainente, F., Zapparoli, G., Cecconi, D., & Simonato, B. (2016b). Food Chemistry, 199, 639−647.
- Rousseaux, S., Diguta, C. F., Radoï-Matei, F., Alexandre, H., & Guilloux-Bénatier, M. (2014). Food Microbiology, 38, 104−121.
- Tosi, E., Fedrizzi, B., Azzolini, M., Finato, F., Simonato, B., & Zapparoli, G. (2012). Food Chemisty, 130, 370-375.
- Zapparoli, G., Lorenzini, M., Tosi, E., Azzolini, M., Slaghenaufi, D., Ugliano, M., & Simonato, B. (2018). Food Chemistry, 263, 42–50